AliveCor Kardia Mobile 6L FDA Cleared- Mobile ECG Device, New
Arrives by Fri, Mar 1 Buy AliveCor Kardia Mobile 6L FDA Cleared- Mobile ECG Device, New at
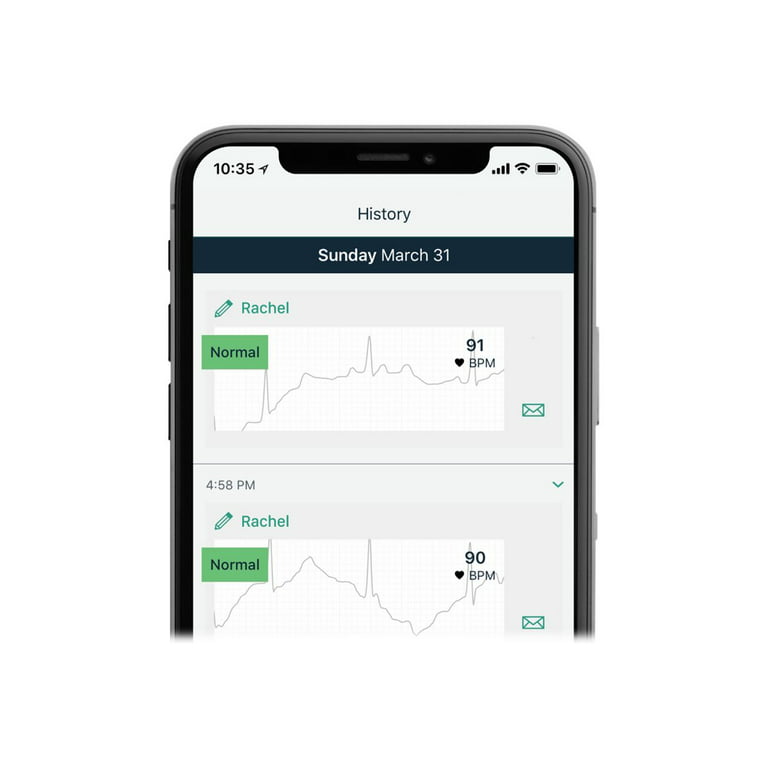
Record medical-grade heart data with KardiaMobile 6L, the world's only six-lead personal EKG. KardiaMobile 6L is FDA-cleared to detect AFib, Bradycardia, Tachycardia, and Normal Sinus Rhythm. Its six-lead heart data gives you and your doctor an in-depth view of your heart rhythm from home. KardiaMobile 6L is the world’s only six‑lead personal EKG. Detect three of the most common arrhythmias in just 30 seconds right on your smartphone. With six leads of heart data, your doctor will have a more complete view of your heart rhythm and can use the insights to help identify other cardiac arrhythmias. You can record a medical-grade, six-lead EKG on your smartphone. You will get an instant analysis of your heart rhythm in just 30 seconds. KardiaMobile is FDA-cleared to detect 3 of the most common arrhythmias. - Save, share, or print your EKGs. - KardiaMobile 6L delivers EKG leads I, II, III, aVL, aVR, and aVF. - No wires, patches, or gels are required. - Works with most smartphones or tablets on Android or iOS.

AliveCor KardiaMobile 6-Lead ECG Monitor

AliveCor KardiaMobile 6L ECG, FDA-Cleared, Wireless 6-Lead EKG

Alivecor Kardiamobile 6L Personal EKG Device that detects AFib

FDA Grants First Ever Clearance For Six-Lead Personal ECG Device
Kardia on the App Store

AliveCor - KardiaMobile 6L Personal EKG Monitor - Black

ALIVECOR KARDIAMOBILE 6L MOBILE 6-LEAD EKG BRAND NEW FACTORY SEALED 850214007148

IronRod Health - How To: Record Your EKG Using the KardiaMobile 6L in the Omron App

Kardia Mobile 6L

EKG Monitor with 6 Leads KardiaMobile 6L by AliveCor – AliveCor, Inc.

KardiaMobile 6L – AliveCor EU
ACCURATE AND DETAILED EKG RESULTS. KardiaMobile 6L records a medical-grade, six-lead EKG and provides FDA-cleared determinations of your heart rhythm

KardiaMobile 6-Lead Personal EKG Monitor – Six Views of The Heart – Detects AFib and Irregular Arrhythmias – Instant Results in 30 Seconds – Works

ALIVECOR KARDIAMOBILE 6L MOBILE 6-LEAD EKG BRAND NEW FACTORY SEALED 850214007148

FDA Clears AliveCor's KardiaMobile 6L as First Six-Lead Personal ECG Device









