FDA warns of risk of overheating, fire with some FreeStyle Libre glucose monitors

Lithium batteries pose risks for wearable health devices. #CCL Connected Energy Solutions, Michael Crane posted on the topic

Freestyle Libre Recall Issued For 4.2M Glucose Monitors After Reports of Fires, Extreme Heat

Diabetes Dialogue: News on Abbott Readers & CMS Expansion of CGM Availability

MPO's Most-Read Stories This Week—April 15

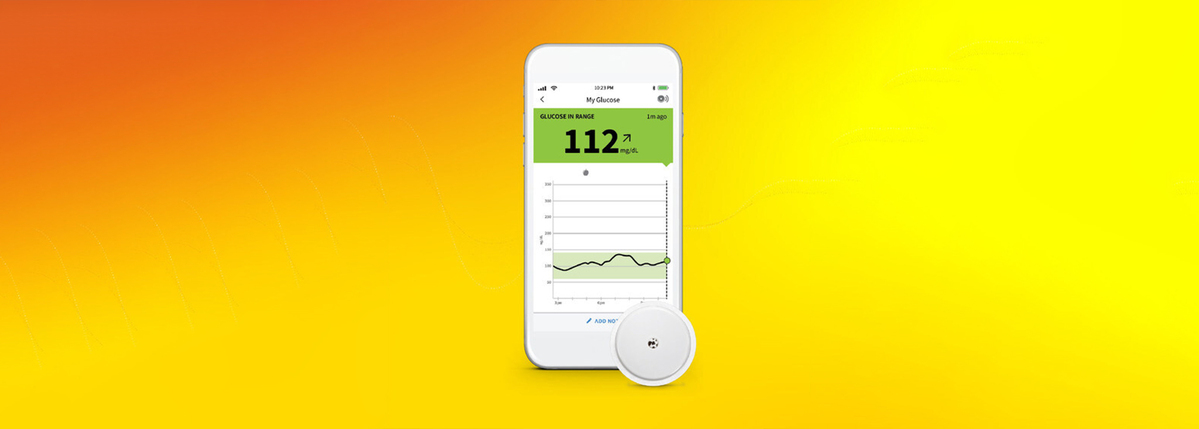
Abbott's Freestyle Libre system becomes first CGM to be FDA cleared for use without fingersticks

Abbott's FreeStyle Libre Flash Glucose Monitoring Systems Recalled Due to Extreme Heat and Fire Hazards - Parker Waichman LLP
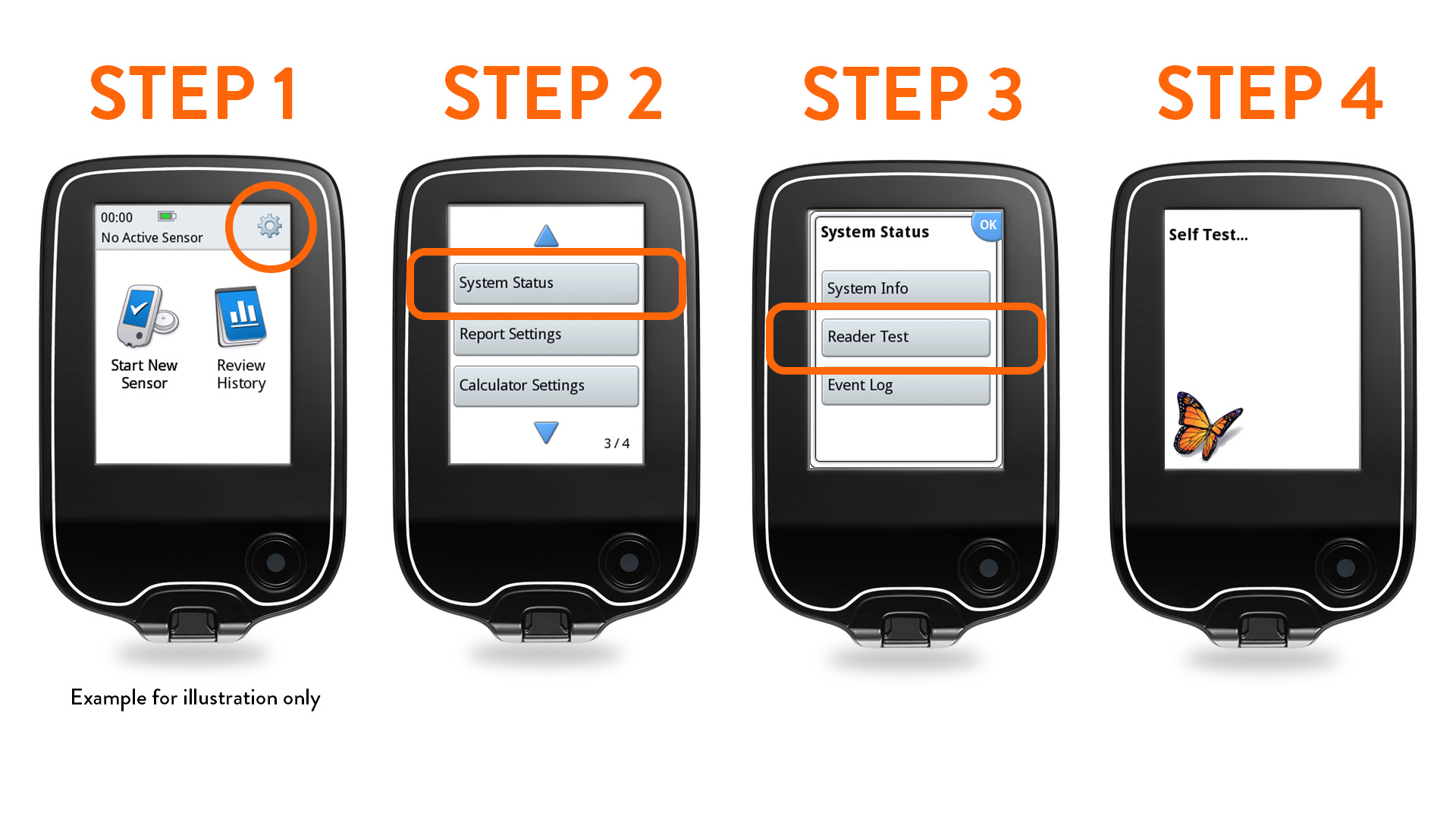
FAQs FreeStyle - Abbott

FDA warns against using smartwatches and smart rings to measure blood sugar
FreeStyle Libre 2 Flash Blood Glucose Monitoring System — Mountainside Medical Equipment

FDA warns of risk of overheating, fire with some FreeStyle Libre glucose monitors


(5).jpg)